Ground Beef and Culture-independent and Spoilage
Introduction
Beef is 1 of the virtually commonly consumed meats worldwide, including Korea (Cho et al., 2010), withal, beefiness products are highly perishable (Doulgeraki et al., 2012). Microorganisms in beefiness can crusade the spoilage of products and food poisoning. Since beef meat is food rich and has high water content, microorganisms from the processing environments can easily colonize beef meat (De Filippis et al., 2013). Even during storage in refrigeration temperatures, psychrotrophic leaner such as lactic acrid bacteria and Pseudomonas spp. can grow on beef meat, thereby increasing the risk of meat spoilage (Doulgeraki et al., 2012). In addition, outbreaks due to contamination of beef meat with Escherichia coli O157 and Salmonella spp. accept continuously occurred despite the maintenance of loftier hygiene levels (Kivi et al., 2007; Friesema et al., 2012; Heiman et al., 2015). Several studies have analyzed spoilage leaner and pathogens by culture-based methods to find ways to reduce spoilage and foodborne affliction (Ercolini et al., 2006; Black et al., 2010; Limbo et al., 2010). Recent studies using loftier-throughput sequencing methods have also reported the presence of meat spoilage-related microorganisms and pathogens during processing steps or under different storage conditions (De Filippis et al., 2013; Hultman et al., 2015; Stoops et al., 2015; Yang et al., 2016). However, these studies focused on specific bacteria and provided limited information on the overall microbial composition of beef meat. Therefore, analyzing the whole microbiota associated with beef is essential to understanding the spoilage gamble of fresh beef meat.
Furthermore, understanding the potential role of microbes in fresh beef meat before farther processing is too important, since it is the initial status of microbiota and tin influence the alteration of beef microbiota during further processes. Microorganisms more often than not interact with each other to maintain their functions (Freilich et al., 2010; Faust and Raes, 2012; Zheng et al., 2015). These interactions betwixt microbes could be related to the spoilage in food products. Previous studies have also reported that two or more microorganisms contributed to spoilage simultaneously by interacting with each other (Borch et al., 1996; Jørgensen et al., 2000).
This study aimed to analyze the microbiota composition in fresh beef meat and their potential functions influencing the spoilage of meat and the alteration of microbiota during further processing. We compared the microbiota of fresh beefiness meat (ground and non-ground) collected from different sites in unlike seasons in Republic of korea by using the Illumina MiSeq sequencing. The effects of the environmental variables on bacterial distributions in beef meat were analyzed, and the spoilage risk was predicted using the information gathered from the results. The outcomes of the nowadays study provide insights into initial microbiota in fresh beefiness meat and extended our understanding of spoilage by the microbiota in beef meat.
Materials and Methods
Sample Collection
A full of 120 beefiness samples (60 non-basis and 60 ground samples) were collected from the Livestock Processing Centre (LPC; the government local livestock joint market) from three different sites (Supplementary Figure S1). These sites were the areas with maximum beef production in Korea based on the almanac report of livestock production and marketing channelone. Cattle from different farms were gathered at the LPC of each site and candy, including slaughter. The beef meat was transferred to market or visitor for further processing. Therefore, the microbiota in beef meat from the LPC is an initial status of microbiota in fresh beef meat. To determine the influence of seasonal differences on beef microbiota, we collected the samples in January and July 2018. Four kilograms of the beef sample (x non-footing and 10 footing samples at each site) were collected and transported in an ice box to the laboratory. Samples were stored at −80°C until further experiments.
Metagenomic DNA Extraction
Non-ground beefiness was cutting into 5 g cubes, and 5 cubes were selected randomly. Footing beef was homogenized, and 25 k of sample was randomly selected. The samples were diluted in 225 mL of buffered peptone water (10 one thousand peptone, 5 yard sodium chloride, three.five k disodium phosphate, and 1.5 1000 potassium dihydrogen phosphate; at pH 7.ii). Bacterial cells were detached from beefiness using a spindle (microorganism homogenizer, Korea patent registration x-2010-0034930) and stored at −80°C. Metagenomic Dna was extracted from each sample using the phenol Deoxyribonucleic acid extraction method previously described (Lee et al., 2016). Extracted genomic DNA was purified with the PowerClean DNA Clean-upward kit (MO Bio Laboratories, Carlsbad, CA, Usa) and confirmed through 1% agarose gel electrophoresis.
Qunatitative Real-Time Polymerase Chain Reaction
The bacterial amounts in each sample was estimated by quantitative real-time PCR of 16S rRNA genes equally previously described (Lee et al., 2017; Kim et al., 2019). The amplification was performed using primers 340F (5′-TCC TACGGGAGGCAGCAG-iii′) and 518R (5′-ATTACCGCG GCTGCTGG-iii′) on a Thermal Cycler Dice Real Time System 3 (Takara Bio, Otsu, Japan). Triplicate reactions of each sample were conducted in a final volume of 25 μL containing 12.5 μL of 2 × SYBR Dark-green PCR primary mix (Bioneer, Korea), ii μM of each primer, and i μL of a DNA template (10-fold dilution serial of sample Deoxyribonucleic acid) or distilled water (negative control) under the post-obit weather: 94°C for v min, followed by forty cycles of denaturation at 94°C for 30 south, annealing at 55°C for 30 s, extension at 72°C for xxx s, and final extension at 72°C for ten min. Standard curves were generated from parallel PCRs of serial log-concentrations (one × 102–one × 108) of 16S rRNA factor copy numbers of the E. coli K12 w3110 strain. Regression coefficients (r ii) for all standard curves were ≥0.99. Differences between samples were adamant with the Isle of mann–Whitney U-exam using R software ver.three.2.0. Values of p < 0.05 were considered statistically significant.
MiSeq Sequencing
The extracted Deoxyribonucleic acid was amplified using primers (targeting V1-V3 region of the 16S rRNA gene) with adapters (forrad: 5′-adapter [TCGTCGGCAGCGTCAGATGTGT ATAAGAGACAG]-GAGTTTGATCMTGGCTCAG-3′; reverse: v′-adapter [GTCTCGTGGGCTCGGAGATGTGTATAAGAG ACAG]-ATTACCGCGGCTGCTGG-3′). PCR distension followed grooming of a 16S metagenomics sequencing library for the MiSeq system (Illumina, Inc., San Diego, CA, United states) was performed equally described previously (Lee et al., 2017; Kim et al., 2019). The library was quantified using a PCR Thermal Cycler Dice Real-Time System Iii (Takara Bio.) with the GenNext NGS Library Quantification Kit (Toyobo, Osaka, Japan). Equimolar concentrations of each library from the different samples were pooled and sequenced using an Illumina MiSeq organization (300 bp-paired ends), following the manufacturer's instructions.
Sequencing Data Analysis
The obtained sequences were analyzed using CLC genomic workbench (ver.11.0.1) with the Microbial Genomics Module (Qiagen) as previously described (Lee et al., 2017; Kim et al., 2019). Paired sequences were merged, and low-quality sequences (<430 bp of merged reads or quality score <xxx) and chimeric reads were removed using the USEARCH pipeline v.10.0.2402. Primer sequences were removed from the merged sequences, and sequences were clustered into operational taxonomic units (OTUs) by 97% sequence identity with the EzTaxon-e database (Yoon et al., 2017). The representative sequences in each OTU cluster were identified, their taxonomic position based on the EzTaxon-due east database. To compare variety indices, the numbers of reads in each sample were normalized by random subsampling and indices were calculated using MOTHUR (Schloss et al., 2009). Differences between samples were evaluated with the Isle of man–Whitney U-examination and Kruskal–Wallis exam in the R software. Results with p < 0.05 were considered statistically significant.
Canonical correspondence analysis (CCA) was performed to analyze the factors influencing the composition of microbiota using the Bray-Curtis distance matrix in the R software, while the significance was evaluated with the permutation test. To find the cadre genus in beef meat, the relative abundance of genera in each sample was used with the Venn package in R. The relative abundance of genera among samples was compared by the heat map using the pheatmap package in R. For this analysis, the genus with over ane% relative abundance in each sample was selected. Benjamini-Hochberg FDR was used to right for multiple tests. Effect with FDR < 0.01 were considered statistically significant.
Prediction of Microbiota Function
The potential function of each group was predicted by the phylogenic investigation of communities by reconstruction of unobserved states ii(PICRUSt2) (Douglas et al., 2019). The abundance of the predicted function was normalized concerning 16S rRNA gene copy numbers, and MetaCyc pathways were used for analyzing predicted functions of microbiota. The statistical differences betwixt groups were determined using the two-sided Welch's test, and confidence intervals were calculated using the Welch'southward inverted examination in the Statistical Analysis of Metagenomic Profiles (STAMP) software (Parks and Beiko, 2010). Benjamini-Hochberg FDR was used to right for multiple tests. Just pregnant results with q-value (corrected p < 0.01) were used.
Results and Discussion
Comparing of Diversity Indices and Bacterial Compositions Amongst Samples
A total of 7,551,419 reads (average of 41,475 reads for Jan samples and 84,382 reads for July samples) were analyzed after the trimming process for the 120 beefiness samples (Supplementary Table S1). The numbers of observed OTUs were higher in the samples collected in July (average 61,041 ± 7,843) than in January (24,559 ± 1,922; p < 0.0001). The number of observed OTUs was highest in the footing beef from sampling site C in July (JulCG, 81,176 ± 39,019) and lowest in not-ground beef from site A in Jan (JanAnG, 11,415 ± 3,005). The Shannon diversity indices were compared between samples collected in January and July, as well as between samples from dissimilar sites at the same fourth dimension. The average diverseness of the microbiota was higher in samples collected in January (3.63 ± 0.12) than in July (2.73 ± 0.11) (p < 0.0001; Figure 1A). For January samples, the highest variety was detected in non-basis beefiness from site B (four.44 ± 0.08), and the lowest diverseness was detected in not-footing beef from site A (2.54 ± 0.25; p < 0.0001) (Figure 1B). For July, not-ground beef from site A had the highest diverseness (iii.20 ± 0.22), while not-ground beef from site C had the lowest diversity (1.99 ± 0.34; p < 0.01) (Figure 1C).
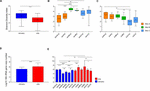
Figure 1. Comparison of bacterial diversity and abundance in beefiness meat samples obtained from different sites and times. (A) Comparison of bacterial diversity between samples obtained in January and July. (B) Comparing of diversity indices of samples obtained in Jan and (C) July. (D) The average bacterial abundance was compared between samples obtained in Jan and July. (Due east) The bacterial abundance in each grouping was compared. The sample proper noun indicates the sampling month, site, and processing types (for example, JanAnG; sample nerveless in January from site A and non-ground beef). Fault bars indicate the standard departure. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
The relative bacterial abundance was determined and compared amongst samples using quantitative real-fourth dimension PCR. The abundance of bacteria in beef samples was higher in July (average 1.45 × 10half-dozen copies/g) than in Jan (boilerplate 1.44 × xv copies/one thousand; p < 0.0001) (Figure 1D). The highest bacterial abundance was detected in ground beef from site B in July (average 2.39 × 10seven copies/g), while the lowest bacterial affluence was detected in not-footing beef from site B in January (average one.23 × 104 copies/thou) (Figure 1E). These results indicate that the decreased diversity of July samples with higher bacteria affluence could be considering of the say-so of some bacteria in the microbiota.
The composition of microbiota in beefiness samples was compared at phylum and genus levels (Figure 2). Firmicutes (51.03%) and Proteobacteria (36.58%) were the ascendant phyla in all beef samples. The proportions of Firmicutes were higher in July (boilerplate 63.xx%) than in January (average 38.86%) (p < 0.05). Betwixt the sites, site B revealed college relative affluence of Actinobacteria both in January (boilerplate fourteen.93%) and July (average 29.67%) samples. However, the identified genera were more diverse in Jan than in July samples (Figures 2C,D). Further, the composition of the microbiota differed between samples nerveless from different sites in January. The dominant genera in samples from site A were Pseudomonas, Carnobacterium, and Brochothrix, while the dominant genera in samples collected from site B were Serratia, Kocuria, and Corynebacterium and in those collected from site C, Escherichia, Macrococcus, and Salmonella. In July samples, Carnobacterium (average 28.xi%), Lactobacillus (average 19.49%), and Pseudomonas (average fourteen.54%) were dominant in all samples. However, Serratia and Kocuria were dominant simply in samples of site B, similar the microbiota in Jan samples. Carnobacterium is a prevalent member of lactic acid leaner (LAB) in fresh meat and processed meat products (Pothakos et al., 2015). Psychrotrophic leaner such every bit LAB and Pseudomonas spp. can easily dominate in meat products stored at under chilled conditions (Stanborough et al., 2017). Although the average temperature in July at the three sites was above 26°C (27.ii°C at site A, 28.three°C at site B, 26.7°C at site C), psychrotrophic bacteria were dominant in July. This could exist considering of the common cold temperatures (beneath 10°C) during processing in the LPC and during transportation weather. Though the cold temperature in the LPC and transportation was also maintained in January, the more than diverse microbiota in January could be because of the storage duration of beef meat. An earlier study has reported higher bacterial diversity in fresh-cutting beef than the later on phase of storage (Säde et al., 2017).
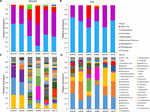
Figure 2. The limerick of microbiota was compared among samples at the phylum and genus levels. Comparison of phylum composition in beef meat nerveless in (A) January and (B) July. Comparison of genus composition collected in (C) January and (D) July. Taxa with relative abundance <ane% in each sample were combined with the "other" group.
In addition, the dominant genera differed past sampling sites. Serratia was peculiarly dominant in ground beef from site B in both months (26.32% in January and 24.0% in July). Serratia spp. is commonly found in meat (Doulgeraki et al., 2011) and is too known as a major spoilage Enterobacteriaceae (Ercolini et al., 2006). Withal, Escherichia (16.32% in ground beefiness) and Salmonella (fifteen.22% in not-ground beef) were ascendant in beefiness samples from site C in January. The observed differences of microbiota could be because of the environments of the LPC, farm, and individual differences of microbiota in cattle. Cattle from different farms were gathered to the LPC of each site; thus, the microbiota in cattle was already dissimilar before slaughter. Nevertheless, cattle were washed and processed nether a controlled environment in the LPC. Therefore, the microbiota in beef meat could be influenced more by the surroundings of the LPC, implicating its importance for nutrient rubber. The findings of this study were supported by earlier studies, showing the importance of the processing environment for food safety and reducing microbial contagion (Rivera-Betancourt et al., 2004; Nychas et al., 2008; Stellato et al., 2016).
Factors Influencing the Differences in Microbiota in Dissimilar Beef Samples
Canonical correspondence analysis was used to bear witness the correlation of microbiota divergence with environmental variables (Figure 3A). The full inertia of the CCA plot was 5.88, and the constrained inertia was 1.13. A full of 8.6% of the constrained inertia was explained by the CCA1 axis, while CCA2 explained a farther 5.6%. Arrows on the plot showed the forcefulness of the plot dispersion. Amid the arrows, sampling sites and months had a more than significant influence on microbiota dispersion than the processing blazon. Microbiota in January samples were more than distinguished according to sampling sites than those in July samples. For July, samples from site B were significantly different from the other two sites. This difference was also observed in the genera composition of samples from site B (Figure 2d). We found that beef microbiota were significantly dissimilar in the ground samples from site B and C in Jan. In addition, the cluster dendrogram likewise showed that the altitude between basis beef and non-ground beefiness was relatively high in these sites, while other samples shared similar communities irrespective of the processing types (Figure 3B). The CCA plot indicated that the regional and seasonal factors comprehensively afflicted the variety of beef microbiota. Before studies have shown the possibility of transmission of the microbes present in beef, cattle, and the processing machinery to the beef products (Elder et al., 2000; Stellato et al., 2016). From the present findings, we also speculated that the cleanliness of farm and processing environments, water quality, and storage weather of the LPC could have influenced the composition of the microbiota in the beefiness products. Hence, at that place is a demand for careful process management before, during, and after slaughtering.

Figure 3. The contrast of microbiota in beef meat samples. (A) Canonical correspondence analysis (CCA) biplot of beef microbiota. The points indicate the microbiota of each sample, and the dissimilarity was calculated by Bray-Curtis distance. The axes correspond the percentage of the corresponding total variance. The variables (sampling months, sites, and processing types) were depicted every bit vectors. The longer vector has stronger effects on the dispersion of microbiota. (B) Clustering of microbiota based on Bray-Curtis altitude between samples.
The genera in the samples nerveless from different sites at different months subjected to different grinding processes were determined by DeSeq2 based on the log2 fold change values of genus proportion (Supplementary Table S2). Average of 14 genera for non-ground and 15 genera for ground beef were found to be significantly unlike between sampling months in each sampling site (FDR < 0.01). Propionibacterium was dominant in January samples from all sites (5.62 ± 0.seventy-fold), while Serratia (iv.71 ± 0.27-fold), Hafnia (half dozen.39 ± 0.77–fold), Lactobacillus (5.26 ± 0.25–fold), and Lactococcus (12.42 ± 7.46-fold) were ascendant in July samples. The dominance of Propionibacterium in Jan samples could exist because of contamination from human skin and indoor slaughter environments (Jeon et al., 2013; Stoops et al., 2015; Alessandria et al., 2016). However, a reduced level of Propionibacterium in the July sample could be for the dominance of LAB, such as Lactobacillus and Lactococcus. Reported earlier, these LAB could inhibit the growth of Propionibacterium acnes through the secretion of bacteriocins (Oh et al., 2006; Kober and Bowe, 2015). In add-on, LAB play a major role in beef spoilage even at refrigerated temperatures (Nychas and Skandamis, 2005), and the abundance of LAB, Serratia, and Hafnia genera in the present study could be related to the beef spoilage. These results indicated that the environmental conditions in July favor spoilage; hence, management of beef meat should be paramount in July for reducing risk of spoilage.
The ground samples nerveless from the 3 sites in January and July identified vii–19 significantly different genera (FDR < 0.01), however, we did not notice any significant departure between the basis and not-ground beef from sites A and C in July (FDR > 0.01). In addition, the cluster dendrogram showed that the similarity of microbiota between basis and not-basis beef from the aforementioned sites in each month was relatively loftier (Figure 3B). The findings of this study indicated that the ground procedure is non a factor influencing the microbiota in beef meat.
The Core Genera in Beef Microbiota in All Samples
Based on the results of the cluster dendrogram that showed the minimal influence of the ground process on the community dissimilarity (Effigy 3B), nosotros determined the core genera in the beefiness samples collected from different sampling sites and times (Supplementary Figure S2). The identified core genera are summarized in Supplementary Tabular array S3. A total of 52 genera were detected as core genera amongst all beef samples with 27 genera in January and 25 genera in July samples.
Afterwards, the relative abundances of core genera were compared amidst samples through oestrus map analysis (Effigy 4). The samples were clustered into four groups based on Spearman correlation, and groups were distinguished by the sampling month (Jan samples in groups 1 and 4; July samples in groups 2 and 3). We then classified the genera into four graphic symbol groups related to potential risk factors according to the previous studies (Nychas et al., 2008; Doulgeraki et al., 2012; Iulietto et al., 2015). The groups were characterized as common (unremarkably found genera in fresh beef), spoilage (genera related to beef spoilage), pathogen (related to potential foodborne pathogens), and NR (not reported). We identified eight genera every bit common, 12 genera as spoilage, five genera as pathogen, and 17 genera as NR (Tabular array 1). In this report, the relative affluence of spoilage genera (63.eleven%) in beefiness microbiota was highest (p < 0.001), followed by NR (12.61%), mutual (eleven.17%), and pathogen (7.71%). Nevertheless, 84.59% of the core genera were Firmicutes and Proteobacteria and mostly comprised the spoilage group that included eight genera of Firmicutes, 3 genera of Proteobacteria, and one genus of Actinobacteria. These results were consistent with previous studies, which have reported Firmicutes and Proteobacteria as the dominant phyla in spoiled beef (Vihavainen and Björkroth, 2007; De Filippis et al., 2013).
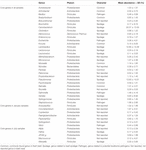
Table ane. Core genera in the microbiota of beef meat. Genera were classified into 4 character groups according to the previous studies (Nychas et al., 2008; Doulgeraki et al., 2012; Iulietto et al., 2015).
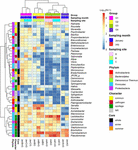
Effigy 4. Comparison of core genera using heat map analysis. The relative abundances of genera were calculated by a log10 calibration. Samples were clustered to groups 1, 2, 3, and 4 by spearman correlations. Cadre genera were characterized by mutual, pathogen, spoilage, and NR.
The genus Salmonella belonging to the group "pathogen" was constitute to be more arable in beef samples from site C in January than the other three groups. Information technology has been shown that Salmonella spp. are the virtually common pathogens causing foodborne disease related to beef consumption (Dechet et al., 2006; Laufer et al., 2015). A previous study reported that S. enterica was more abundant in final beef products than in the feedlot (Yang et al., 2016). Although diverse pathogens tin be eliminated by the awarding of surface antimicrobial treatments, Salmonella spp. can survive by internalization into peripheral lymph nodes and multiply (Brichta-Harhay et al., 2012).
The relative abundances of genera related to spoilage were higher in the samples of grouping ii, three, and 4 (lx.84, 82.82, and 84.90% of core genera, respectively). The risk of spoilage might be higher in the beefiness samples of these groups. Although the relative abundances of Carnobacterium and Pseudomonas were high in these beef samples, the proportions of spoilage genera were different among samples in these groups. Serratia was the dominant genus in grouping 2 (19.05%), Lactobacillus in grouping 3 (27.05%), and Brochothrix in group 4 (22.86%). These differences could exist because of the existence of unlike microbiota in different samples and the influence of the ecology conditions of the respective LPC. The findings of this written report reveal that the composition of microbiota in beef meat could provide the data for microbial risks related to spoilage.
The Predicted Function of the Beef Core Microbiota
Comparison of the predicted functions of microbiota betwixt samples in group 1 and other groups using PICRUSt2 (Effigy 5) identified 221 significantly different pathways between group ane and other groups (FDR < 0.01). The details of the groups with over 0.20% difference are provided in Figure 5 and Supplementary Table S4.
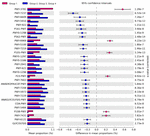
Figure 5. Comparison of predicted pathways betwixt group i and the other groups clustered in heat map assay (Figure 4). Pathways were predicted past PICRUSt2. The significantly different pathways betwixt groups (Welch'due south t-test q < 0.01) are shown.
The pathways related to the TCA bike and aerobic respiration were significantly prominent in the samples of group 1 than in the other groups (Supplementary Tabular array S4). These results indicated that the aerobic bacteria were predominant and played important roles in the samples of group 1. Nosotros also observed higher abundance of Salmonella in these samples. The results were consistent with a previous study that showed the affluence of Salmonella corresponded to the affluence of aerobic bacteria in beef (Brichta-Harhay et al., 2008). Here, the proportions of pathways related to fermentation and glycolysis were significantly college in samples of groups 2, 3, and 4. These results indicated that fermentation may be the primal pathway leading to beefiness spoilage. Homolactic fermentation (ANAEROFRUCAT-PWY) and pyruvate fermentation to acetate and lactate II (PWY-5100) pathways were significantly college in the samples of groups 2, 3, and iv than in the samples of group 1. These pathways were also related to beefiness souring in a previous written report (Nychas et al., 2008). Lactate can maintain a low pH in beef meat; thus, bacteria with acid-tolerance tin can still thrive and dominate in such surroundings (Alvarado and McKee, 2007; Kalchayanand et al., 2018). The proportions of glycolysis and sucrose degradation pathways were significantly higher in the samples of groups ii, iii, and 4. Glucose is ane of the chief precursors for off-flavors and acids by spoilage microbes such as Carnobacterium, Brochothrix, LAB, and Pseudomonas (Gram et al., 2002). In addition, Lactobacillus tin can cause astringent acidification in beef and emission of off-aroma compounds, while Leuconostoc tin can produce organic acid such as acerb acid by using glucose in spoiled beef (Samelis et al., 2006; Doulgeraki et al., 2010; Pothakos et al., 2015). Therefore, spoilage genera in the samples of groups 2, 3, and 4 could dominate past producing acid and, as a result, could be related to beef spoilage in beef meat.
Microbiota in samples of group 1 were more than diverse than those of other groups, and the relative abundances of aerobic bacteria were higher in the microbiota of group 1. In contrast to microbiota in the samples of group i, the microbes related to the spoilage were more than detected in the microbiota of groups 2, three, and four samples, even at cold temperatures. These leaner may inhibit the aerobic bacteria through fermentation in beef meat. Therefore, beefiness meat may be spoiled. The initial contaminating microbes and the storage status were of import to the afterward stage microbiota in beef (Hilgarth et al., 2018), and spoilage could occur past metabolites produced past spoilage microbes (Jääskeläinen et al., 2016). In this study, the potency of spoilage microbes may be related to pathways of glucose utilization, and it could crusade beefiness meat spoilage by resident microbiota.
In this report, we analyzed the microbiota in fresh beefiness meat and their potential functions past microbiota characteristics. The microbiota in fresh beef meat differed according to sampling sites and months, simply core genera were detected in all samples. The potential spoilage genera were prominent in fresh beef meat, and these genera could influence the growth of other microbes using glucose by fermentation. Beef meat has a glucose-enriched environment; thus, strategies to inhibit the spoilage microbes using obtained information can reduce and preclude the spoilage of beef meat by microbiota. Although further studies such as co-culturing, metatranscriptomic, and metabolomics analyses are necessary to validate the results, the findings in this study provide information on initial microbiota to understand the bacterial risk of spoilage in beef meat products.
Data Availability Statement
All sequences were deposited on European Nucleotide Archive (ENA) study accession number PRJEB35021 (https://www.ebi.air-conditioning.u.k./ena/data/search?query=PRJEB35021).
Author Contributions
SC and B-SK: conception and design. BH and HC: acquisition of information. BH: information analysis and writing of the draft manuscript. BH and B-SK: data interpretation. BH, HC, SC, and B-SK: review and editing the manuscript. All authors contributed to the commodity and approved the submitted version.
Funding
This piece of work was supported by the Ministry of Food and Drug Prophylactic, Southward Korea (20162MFDS142), and by the National Enquiry Foundation of Korea, funded by the Ministry building of Science, ICT, and Future Planning (2017R1E1A1A01074639).
Disharmonize of Interest
The authors declare that the enquiry was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Cloth
The Supplementary Fabric for this article can be establish online at: https://www.frontiersin.org/articles/10.3389/fmicb.2020.01657/total#supplementary-fabric
Footnotes
- ^ http://www.ekapepia.com
- ^ http://world wide web.drive5.com/usearch
References
Alessandria, V., Ferrocino, I., De Filippis, F., Fontana, G., Rantsiou, K., Ercolini, D., et al. (2016). Microbiota of an Italian grana-like cheese during manufacture and ripening, unraveled by 16S rRNA-based approaches. Appl. Environ. Microbiol. 82, 3988–3995. doi: 10.1128/AEM.00999-16
PubMed Abstract | CrossRef Full Text | Google Scholar
Alvarado, C., and McKee, S. (2007). Marination to ameliorate functional properties and safety of poultry meat. J. Appl. Poult. Res. sixteen, 113–120. doi: x.1093/japr/16.1.113
CrossRef Total Text | Google Scholar
Blackness, Eastward., Hirneisen, 1000., Hoover, D., and Kniel, M. (2010). Fate of Escherichia coli O157: H7 in footing beefiness following high-pressure level processing and freezing. J. Appl. Microbiol. 108, 1352–1360. doi: 10.1111/j.1365-2672.2009.04532.x
PubMed Abstruse | CrossRef Total Text | Google Scholar
Borch, E., Kant-Muermans, K.-L., and Blixt, Y. (1996). Bacterial spoilage of meat and cured meat products. Int. J. Food Microbiol. 33, 103–120. doi: 10.1016/0168-1605(96)01135-X
CrossRef Full Text | Google Scholar
Brichta-Harhay, D. Thousand., Arthur, T. K., Bosilevac, J. M., Kalchayanand, N., Schmidt, J. Westward., Wang, R., et al. (2012). Microbiological analysis of bovine lymph nodes for the detection of Salmonella enterica. J. Food Prot. 75, 854–858. doi: 10.4315/0362-028X.JFP-11-434
PubMed Abstract | CrossRef Full Text | Google Scholar
Brichta-Harhay, D. M., Guerini, M. N., Arthur, T. Grand., Bosilevac, J. K., Kalchayanand, North., Shackelford, South. D., et al. (2008). Salmonella and Escherichia coli O157: H7 contamination on hides and carcasses of cull cattle presented for slaughter in the United States: an evaluation of prevalence and bacterial loads past immunomagnetic separation and direct plating methods. Appl. Environ. Microbiol. 74, 6289–6297. doi: 10.1128/AEM.00700-08
PubMed Abstract | CrossRef Full Text | Google Scholar
Cho, S., Kim, J., Park, B., Seong, P., Kang, Yard., Kim, J., et al. (2010). Cess of meat quality properties and development of a palatability prediction model for Korean Hanwoo steer beefiness. Meat Sci. 86, 236–242. doi: ten.1016/j.meatsci.2010.05.011
PubMed Abstract | CrossRef Full Text | Google Scholar
De Filippis, F., La Storia, A., Villani, F., and Ercolini, D. (2013). Exploring the sources of bacterial spoilers in beefsteaks by civilisation-independent loftier-throughput sequencing. PLoS One eight:e70222. doi: 10.1371/journal.pone.0070222
PubMed Abstract | CrossRef Full Text | Google Scholar
Dechet, A. M., Scallan, E., Gensheimer, M., Hoekstra, R., Gunderman-King, J., Lockett, J., et al. (2006). Outbreak of multidrug-resistant Salmonella enterica serotype Typhimurium definitive type 104 infection linked to commercial basis beef, northeastern Us, 2003–2004. Clin. Infect. Dis. 42, 747–752. doi: 10.1086/500320
PubMed Abstract | CrossRef Full Text | Google Scholar
Douglas, G. G., Maffei, 5. J., Zaneveld, J., Yurgel, Southward. N., Chocolate-brown, J. R., Taylor, C. Thousand., et al. (2019). PICRUSt2: an improved and extensible arroyo for metagenome inference. BioRxiv [Preprint]. doi: 10.1101/672295
CrossRef Full Text | Google Scholar
Doulgeraki, A. I., Ercolini, D., Villani, F., and Nychas, G.-J. East. (2012). Spoilage microbiota associated to the storage of raw meat in unlike weather. Int. J. Food Microbiol. 157, 130–141. doi: 10.1016/j.ijfoodmicro.2012.05.020
PubMed Abstract | CrossRef Total Text | Google Scholar
Doulgeraki, A. I., Paramithiotis, South., Kagkli, D. M., and Nychas, One thousand.-J. E. (2010). Lactic acid leaner population dynamics during minced beef storage under aerobic or modified temper packaging weather condition. Nutrient Microbiol. 27, 1028–1034. doi: 10.1016/j.fm.2010.07.004
PubMed Abstruse | CrossRef Total Text | Google Scholar
Doulgeraki, A. I., Paramithiotis, S., and Nychas, Thou.-J. East. (2011). Characterization of the Enterobacteriaceae community that developed during storage of minced beef under aerobic or modified atmosphere packaging weather. Int. J. Food Microbiol. 145, 77–83. doi: 10.1016/j.ijfoodmicro.2010.11.030
PubMed Abstract | CrossRef Full Text | Google Scholar
Elder, R. O., Slap-up, J. E., Siragusa, One thousand. R., Barkocy-Gallagher, Chiliad. A., Koohmaraie, K., and Laegreid, West. W. (2000). Correlation of enterohemorrhagic Escherichia coli O157 prevalence in feces, hides, and carcasses of beef cattle during processing. Proc. Natl. Acad. Sci. UsA. 97, 2999–3003. doi: x.1073/pnas.97.7.2999
CrossRef Full Text | Google Scholar
Ercolini, D., Russo, F., Torrieri, Due east., Masi, P., and Villani, F. (2006). Changes in the spoilage-related microbiota of beef during refrigerated storage under unlike packaging conditions. Appl. Environ. Microbiol. 72, 4663–4671. doi: x.1128/AEM.00468-06
PubMed Abstruse | CrossRef Full Text | Google Scholar
Freilich, South., Kreimer, A., Meilijson, I., Gophna, U., Sharan, R., and Ruppin, E. (2010). The large-scale organization of the bacterial network of ecological co-occurrence interactions. Nucleic Acids Res. 38, 3857–3868. doi: 10.1093/nar/qkq118
CrossRef Full Text | Google Scholar
Friesema, I. H., Schimmer, B., Ros, J. A., Ober, H. J., Heck, M. E., Swaan, C. M., et al. (2012). A regional Salmonella enterica serovar Typhimurium outbreak associated with raw beef products, Holland, 2010. Foodborne Pathog. Dis. 9, 102–107. doi: 10.1089/fpd.2011.0978
PubMed Abstract | CrossRef Full Text | Google Scholar
Gram, L., Ravn, L., Rasch, M., Bruhn, J. B., Christensen, A. B., and Givskov, Thousand. (2002). Nutrient spoilage—interactions between nutrient spoilage bacteria. Int. J. Food Microbiol. 78, 79–97. doi: 10.1016/s0168-1605(02)00233-7
CrossRef Full Text | Google Scholar
Heiman, K. Due east., Mody, R. G., Johnson, S. D., Griffin, P. M., and Gould, L. H. (2015). Escherichia coli O157 outbreaks in the U.s., 2003–2012. Emerg. Infect. Dis. 21, 1293–1301. doi: 10.3201/eid2108.141364
PubMed Abstruse | CrossRef Total Text | Google Scholar
Hilgarth, M., Behr, J., and Vogel, R. F. (2018). Monitoring of spoilage-associated microbiota on modified atmosphere packaged beefiness and differentiation of psychrophilic and psychrotrophic strains. J. Appl. Microbiol. 124, 740–753. doi: x.1111/jam.13669
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hultman, J., Rahkila, R., Ali, J., Rousu, J., and Björkroth, K. J. (2015). Meat processing plant microbiome and contamination patterns of cold-tolerant bacteria causing nutrient safe and spoilage risks in the industry of vacuum-packaged cooked sausages. Appl. Environ. Microbiol. 81, 7088–7097. doi: 10.1128/AEM.02228-15
PubMed Abstract | CrossRef Full Text | Google Scholar
Iulietto, One thousand. F., Sechi, P., Borgogni, Eastward., and Cenci-Goga, B. T. (2015). Meat spoilage: a disquisitional review of a neglected alteration due to ropy slime producing bacteria. Ital. J. Anim. Sci. 14:4011. doi: 10.4081/ijas.2015.4011
CrossRef Full Text | Google Scholar
Jääskeläinen, Due east., Hultman, J., Parshintsev, J., Riekkola, M.-L., and Björkroth, J. (2016). Evolution of spoilage bacterial community and volatile compounds in chilled beef under vacuum or high oxygen atmospheres. Int. J. Food Microbiol. 223, 25–32. doi: x.1016/j.ijfoodmicro.2016.01.022
PubMed Abstract | CrossRef Full Text | Google Scholar
Jeon, Y.-Due south., Chun, J., and Kim, B.-S. (2013). Identification of household bacterial community and assay of species shared with man microbiome. Curr. Microbiol. 67, 557–563. doi: 10.1007/s00284-013-0401-y
PubMed Abstract | CrossRef Full Text | Google Scholar
Jørgensen, 50. V., Huss, H. H., and Dalgaard, P. (2000). The consequence of biogenic amine production by single bacterial cultures and metabiosis on cold-smoked salmon. J. Appl. Microbiol. 89, 920–934. doi: 10.1046/j.1365-2672.2000.01196.ten
PubMed Abstract | CrossRef Full Text | Google Scholar
Kalchayanand, N., Arthur, T. K., Bosilevac, J. M., Schmidt, J. W., Shackelford, S. D., Brown, T., et al. (2018). Surface pH of fresh beef every bit a parameter to validate effectiveness of lactic acid treatment against Escherichia coli O157:H7 and Salmonella. J. Nutrient Prot. 81, 1126–1133. doi: 10.4315/0362-028X.JFP-17-469
PubMed Abstract | CrossRef Full Text | Google Scholar
Kim, H. Due east., Lee, J. J., Lee, M. J., and Kim, B. South. (2019). Assay of microbiome in raw chicken meat from butcher shops and packaged products in South korea to observe the potential risk of foodborne disease. Nutrient Res. Int. 122, 517–527. doi: x.1016/j.foodres.2019.05.032
PubMed Abstruse | CrossRef Total Text | Google Scholar
Kivi, M., Hofhuis, A., Notermans, D., Wannet, Due west., Heck, M., Van De Giessen, A., et al. (2007). A beef-associated outbreak of Salmonella Typhimurium DT104 in The Netherlands with implications for national and international policy. Epidemiol. Infect. 135, 890–899. doi: ten.1017/S0950268807007972
PubMed Abstract | CrossRef Full Text | Google Scholar
Laufer, A., Grass, J., Holt, K., Whichard, J., Griffin, P. Yard., and Gould, L. (2015). Outbreaks of Salmonella infections attributed to beef–United States, 1973–2011. Epidemiol. Infect. 143, 2003–2013. doi: x.1017/S0950268814003112
PubMed Abstract | CrossRef Full Text | Google Scholar
Lee, J.-J., Kim, T.-Y., Choi, South. H., and Kim, B.-S. (2017). Analysis of the bacterial microbiome in the small octopus, Octopus variabilis, from S Korea to detect the potential gamble of foodborne illness and to improve product direction. Food Res. Int. 102, 51–threescore. doi: 10.1016/j.foodres.2017.09.084
PubMed Abstract | CrossRef Full Text | Google Scholar
Lee, M.-J., Lee, J.-J., Chung, H. Y., Choi, S. H., and Kim, B.-S. (2016). Analysis of microbiota on abalone (Haliotis discus hannai) in South Korea for improved product direction. Int. J. Food Microbiol. 234, 45–52. doi: ten.1016/j.ijfoodmicro.2016.06.032
PubMed Abstract | CrossRef Full Text | Google Scholar
Limbo, S., Torri, Fifty., Sinelli, Northward., Franzetti, 50., and Casiraghi, Eastward. (2010). Evaluation and predictive modeling of shelf life of minced beef stored in high-oxygen modified atmosphere packaging at unlike temperatures. Meat Sci. 84, 129–136. doi: x.1016/j.meatsci.2009.08.035
PubMed Abstruse | CrossRef Full Text | Google Scholar
Nychas, G. Due east., and Skandamis, P. (2005). "Fresh meat spoilage and modified atmosphere packaging (MAP)," in Improving the Prophylactic of Fresh Meat, ed. J. Due north. Sofos (Cambridge, MA: Woodhead Publishing, Ltd.), 461–502.
Google Scholar
Oh, South., Kim, S.-H., Ko, Y., Sim, J.-H., Kim, M. South., Lee, S.-H., et al. (2006). Consequence of bacteriocin produced past Lactococcus sp. HY 449 on peel-inflammatory leaner. Nutrient Chem. Toxicol. 44, 552–559. doi: 10.1016/j.fct.2005.08.008
PubMed Abstract | CrossRef Full Text | Google Scholar
Pothakos, V., Devlieghere, F., Villani, F., Björkroth, J., and Ercolini, D. (2015). Lactic acid bacteria and their controversial role in fresh meat spoilage. Meat Sci. 109, 66–74. doi: 10.1016/j.meatsci.2015.04.014
PubMed Abstract | CrossRef Full Text | Google Scholar
Rivera-Betancourt, M., Shackelford, S. D., Arthur, T. M., Westmoreland, K. E., Bellinger, G., Rossman, M., et al. (2004). Prevalence of Escherichia coli O157: H7, Listeria monocytogenes, and Salmonella in two geographically distant commercial beefiness processing plants in the U.s.a.. J. Food Prot. 67, 295–302. doi: 10.4315/0362-028x-67.2.295
PubMed Abstract | CrossRef Total Text | Google Scholar
Säde, E., Penttinen, K., Björkroth, J., and Hultman, J. (2017). Exploring lot-to-lot variation in spoilage bacterial communities on commercial modified atmosphere packaged beefiness. Nutrient Microbiol. 62, 147–152. doi: 10.1016/j.fm.2016.ten.004
PubMed Abstruse | CrossRef Total Text | Google Scholar
Samelis, J., BJOeRKROTH, J., Kakouri, A., and Rementzis, J. (2006). Leuconostoc carnosum associated with spoilage of refrigerated whole cooked hams in Greece. J. Food Prot. 69, 2268–2273. doi: 10.4315/0362-028x-69.9.2268
PubMed Abstract | CrossRef Full Text | Google Scholar
Schloss, P. D., Westcott, S. L., Ryabin, T., Hall, J. R., Hartmann, G., Hollister, East. B., et al. (2009). Introducing mothur: open up-source, platform-independent, community-supported software for describing and comparison microbial communities. Appl. Environ. Microbiol. 75, 7537–7541. doi: 10.1128/AEM.01541-09
PubMed Abstract | CrossRef Full Text | Google Scholar
Stanborough, T., Fegan, Due north., Powell, S. Chiliad., Tamplin, One thousand., and Chandry, P. Due south. (2017). Insight into the genome of Brochothrix thermosphacta, a problematic meat spoilage bacterium. Appl. Environ. Microbiol. 83:e02786-sixteen. doi: 10.1128/AEM.02786-xvi
PubMed Abstruse | CrossRef Full Text | Google Scholar
Stellato, Thousand., La Storia, A., De Filippis, F., Borriello, G., Villani, F., and Ercolini, D. (2016). Overlap of spoilage-associated microbiota betwixt meat and the meat processing surroundings in small-scale-scale and large-scale retail distributions. Appl. Environ. Microbiol. 82, 4045–4054. doi: x.1128/AEM.00793-xvi
PubMed Abstract | CrossRef Total Text | Google Scholar
Stoops, J., Ruyters, S., Busschaert, P., Spaepen, R., Verreth, C., Claes, J., et al. (2015). Bacterial community dynamics during cold storage of minced meat packaged under modified atmosphere and supplemented with different preservatives. Nutrient Microbiol. 48, 192–199. doi: 10.1016/j.fm.2014.12.012
PubMed Abstruse | CrossRef Total Text | Google Scholar
Vihavainen, East. J., and Björkroth, K. J. (2007). Spoilage of value-added, high-oxygen modified-atmosphere packaged raw beef steaks by Leuconostoc gasicomitatum and Leuconostoc gelidum. Int. J. Nutrient Microbiol. 119, 340–345. doi: 10.1016/j.ijfoodmicro.2007.08.029
PubMed Abstract | CrossRef Full Text | Google Scholar
Yang, X., Noyes, Due north. R., Doster, E., Martin, J. Northward., Linke, L. M., Magnuson, R. J., et al. (2016). Use of metagenomic shotgun sequencing technology to detect foodborne pathogens within the microbiome of the beef production concatenation. Appl. Environ. Microbiol. 82, 2433–2443. doi: 10.1128/AEM.00078-16
PubMed Abstruse | CrossRef Full Text | Google Scholar
Yoon, S.-H., Ha, Due south.-M., Kwon, S., Lim, J., Kim, Y., Seo, H., et al. (2017). Introducing EzBioCloud: a taxonomically united database of 16S rRNA cistron sequences and whole-genome assemblies. Int. J. Syst. Evol. Microbiol. 67, 1613–1617. doi: x.1099/ijsem.0.001755
PubMed Abstract | CrossRef Total Text | Google Scholar
Zheng, H., Xu, L., Wang, Z., Li, L., Zhang, J., Zhang, Q., et al. (2015). Subgingival microbiome in patients with healthy and ailing dental implants. Sci. Rep. 5:10948. doi: 10.1038/srep10948
PubMed Abstract | CrossRef Full Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fmicb.2020.01657/full
0 Response to "Ground Beef and Culture-independent and Spoilage"
Post a Comment